Reliable profiling of IgG and IgM levels in home-sampled dried blood spots
Published: 2021-10-18
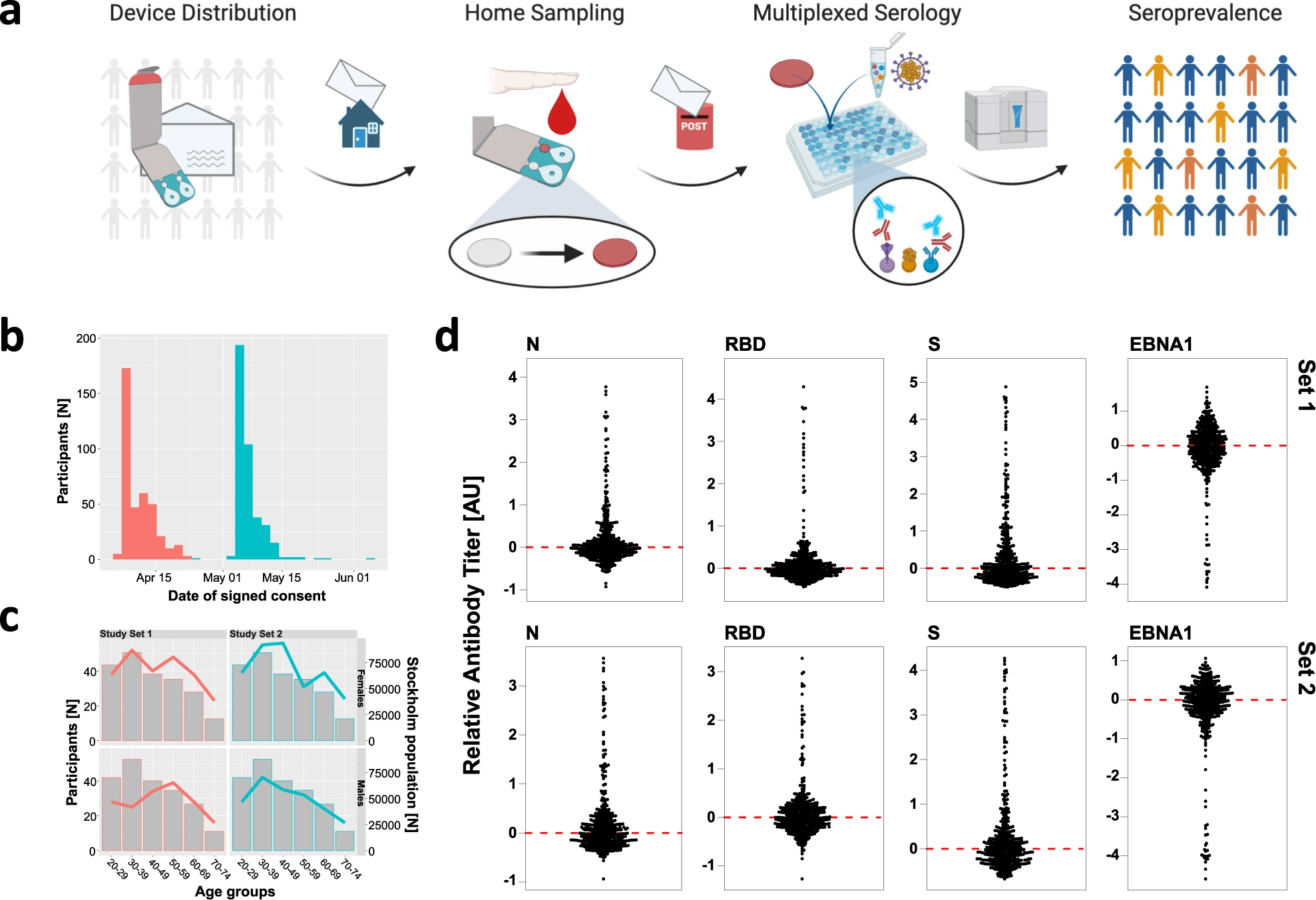
The COVID-19 pandemic has over soon two years challenged societies worldwide in an unprecedented global public health challenge. Different diagnostic methods are important parts of the fight against the SARS-CoV-2 virus and to curb the consequences of the pandemic. Serological assays can be used to study the humoral immune system has coped with a previous infection. However, healthcare professionals are needed to draw venous blood for medical serology testing and the rapid self-tests using capillary blood currently available does not fulfill the requirements. Therefore, home-sampling, by i.e., dried blood spots (DBS), a method which is well-established for screening of neonates, in combination with medical laboratory analysis has been suggested as a route to do serology assays and to decrease societal infections.
In a recent publication in Nature Communications researchers from KTH Royal Institute of Technology, Karolinska University Hospital, Karolinska Institutet, Stockholm University and Danderyd Hospital showed a multianalyte and multiplexed approach to reliably profile IgG and IgM levels against several versions of SARS-CoV-2 proteins (S, RBD, N) in home-sampled dried blood spots (DBS) (First author: Niclas Roxhed, Corresponding authors Jochen M. Schwenk and Niclas Roxhed).
Roxhed and colleagues distributed precision home-sampling kits (two sets of 1000 kits along with questionnaires and forms for informed consent) to randomly selected households in Stockholm during April of 2020. This was during the first wave of the pandemic. They received 55% of the sampling cards back within the first three weeks, and out of these 82% (N = 878) of the participants had succeeded with self-sampling of blood. Women were found to be slightly over-represented in compliance and sampling success. The replies to the questionnaire showed none of the participants had tested positive for SARS-CoV-2 by PCR, though about one in eight (13%) self-reported fever under influenza-like symptoms and one in five (22%) listed symptoms related to issues with breathing. Half of the participants listed no symptoms.
The researchers then analyzed the humoral immune response to SARS-CoV-2 infections in DBS by multiplexed serology assays. The method exhibited a high precision that deemed the utility of using self-sampling of blood for unbiased but reliable estimations of SARS CoV-2 seroprevalence. Roxhed and colleagues used complementary classification approaches to estimate an accumulated seroprevalence of 12.5% (95% CI: 10.3%–14.7%). This includes 5.4% of the samples being IgG+IgM+ against several SARS-CoV-2 proteins, as well as 2.1% being IgG−IgM+ and 5.0% being IgG+IgM− for the virus’ S protein. The results also showed that subjects classified as IgG+ for several SARS-CoV-2 proteins had self-reported influenza-like symptoms more frequently in the questionnaires, than those being IgG+ for only the S protein with odds ratio (OR) = 6.1; p < 0.001. In addition, the study found that about 30% of the seropositive participants were asymptomatic.
In summary, the proposed strategy showed that it was possible to get accurate individual-level and multiplexed serology results from blood collected by self-sampling at home. In addition, the results can contribute to the understanding and knowledge about both the undiagnosed SARS-CoV-2 seroprevalence in society and the diversity of the immune SARS-CoV-2 response. Using home-sampling concepts for health tests can become an important part of future pandemic preparedness to fight future pandemics.
Our project provided unique insights into how the SARS-CoV-2 virus spread during the first wave of the pandemic in the general population in Stockholm. We were excited and thankful that so many participated in our study while accepting that we could not provide any individual feedback from the analyses. Sharing the analysis script and anonymized data is an important aspect of transparent and reproducible research. Hopefully, we will continue to improve the tools, mechanisms and standards that first protect the donor information and then enable to share the collected data.
The article was originally shared on the preprint server medRxiv on July 2nd 2020. The researchers have shared a metadata record-only for the restricted data in the SciLifeLab Data Repository. Roxhed, Niclas; Bendes, Annika; Dale, Matilda; Mattsson, Cecilia; Hanke, Leo; Dodig-Crnkovic, Tea; et al. (2021): Multianalyte serology in home-sampled blood enables an unbiased assessment of the immune response against SARS-CoV-2. SciLifeLab. Dataset. In addition, the authors also state that normalized and anonymized serology data can be made available.
Schwenk and colleagues also shared the analysis scripts used for the articles in GitHub and a record of the scripts was added to the general data repository Zenodo March 26th 2021 under open license.
The researchers within this study were supported by grants to Science for Life Laboratory from the Knut and Alice Wallenberg Foundation (2020.0182) for “Translational Serology”, funds from the Erling-Persson foundation for KTH Center for Precision Medicine (KCAP), KTH, Science for Life Laboratory, and the EU-funded CoroNAb project. SciLifeLab’s Affinity Proteomics Unit in Stockholm, the Autoimmunity and Serology Units, and the Human Protein Atlas Project are acknowledged for their support.
Data
- Metadata record-only for the restricted data: available in SciLifeLab Data Repository
- Analysis scripts: available in GitHub
- Record of the scripts: available in Zenodo
Article
DOI: 10.1038/s41467-021-23893-4
Roxhed, N, Bendes, A, Dale, ma, Mattsson, C, Hanke, L, Dodig-Crnkovic, T, Murray, C, Meineke, B, Elsässer, S, Andréll, J, Havervall, S, Thålin, C, Eklund, C, Dillner, J, Beck, O, Thomas, C.E, McInerney, G, Hong, M-G, Murrell, B, Fredolini, C & Schwenk, J.M. Multianalyte serology in home-sampled blood enables an unbiased assessment of the immune response against SARS-CoV-2. Nature Communications 12, 3695 (2021)